|
10/31/2023 0 Comments Example of electron capture equation
What else is produced here? What else do we make? Well, once again, the number of nucleons is conserved, so I haveĢ34 nucleons on the left, I need 234 on the right. Starting with thorium-234, this nucleus ejects a beta particle, so we go ahead and putĪ beta particle in here, so zero and negative one, If a beta particle isĮjected from the nucleus of a thorium-234, so we're We could put a beta here,Īnd it's an electron, so a negative one charge,Īnd then a zero here. So here's our electron and an electron ejected from the nucleus We saw in the previous video that you represent an electron, since it has a negative one charge, you put a negative one down here, it's not a proton, nor is it a neutron, so we put a zero here. So this is just a visual representation of what's going on here, To eject an alpha particle, so an alpha particle isĮjected from this nucleus, so we're losing this alpha particle, and what's left behind Happening visually, we're starting off with a uranium nucleus which is unstable, it's going The identity of the other product, just look it up here at our table, find atomic number of 90, and you'll see that's thorium here. We already have two positive charges from our alpha particle, and so we need 90 more. On the left, I know I have 92 protons, so 92 positive charges on the left. 
In terms of charge, I knowĬharge is also conserved. Total of 238 on the right, and so therefore nucleonsĪre conserved here. Well, I have four from my alpha particle, so I need 234 more. Trying to figure out the other product from our nuclear equation, I know nucleons are conserved, so if I have 238 nucleons on the left, I need 238 nucleons on the right. So for representing anĪlpha particle in our nuclear equation, since an alpha particle has the same compositionĪs a helium nucleus, we put an He in here, and it has two positive charges, so we put a two down here, and then a total of four nucleons, so we put a four here. Since there are two protons, the charge of an alpha There are two protons in the helium nucleus and two neutrons. 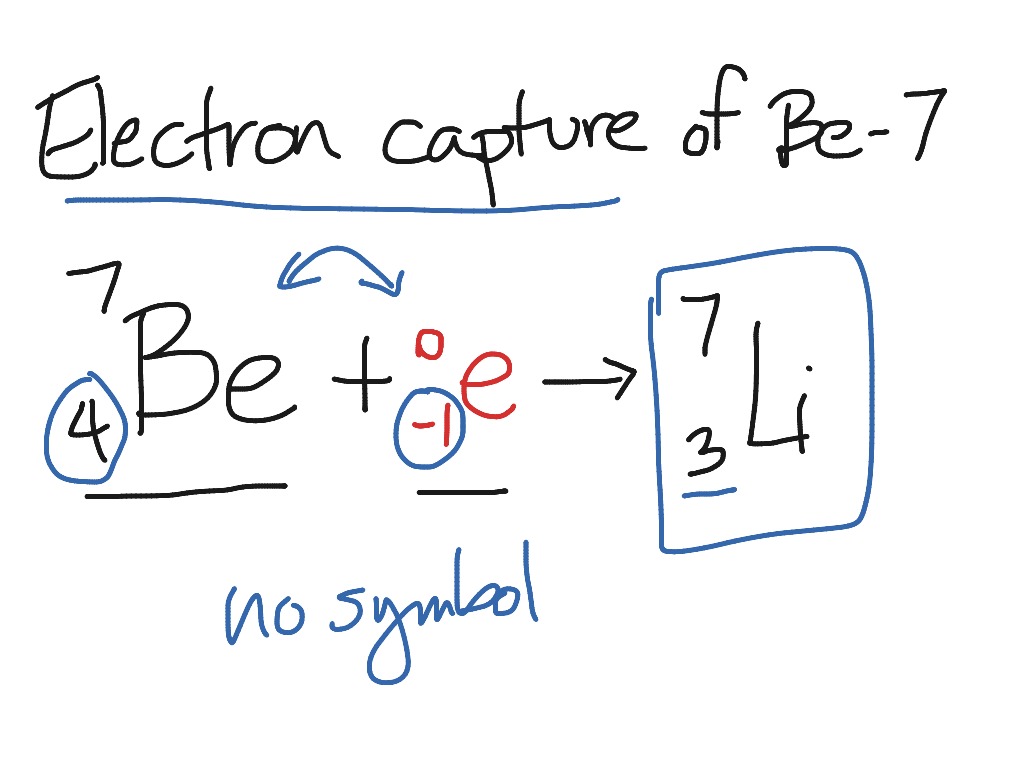
An alpha particle has the same composition as a helium nucleus. In alpha decay, an alpha particle is ejected from an unstable nucleus, so here's our unstable The loss of an alpha particle slightly decreases the p/n ratio (since there are more neutrons than protons in large atoms) as well as decreasing the size of the remaining nucleus.- Let's look at three types of radioactive decay, and we'll start with alpha decay. Electron capture neutralises a proton so increasing the proportion of neutrons. Beta decay increases the proportion of protons, beta plus decay and alpha decay reduce it. 
The ratio within an atom is altered by beta or alpha decay. There is an “ideal” ratio of neutrons to protons for isotopes, dependent on their size, that is shown by the irregular dotted band. The balance of forces and energy within the nucleus is complex. Small atoms can increase their size by fusion. Alpha particles have a mass of 4 amu having 2 protons and 2 neutrons. Large atoms can reduce their size, making them more stable by emitting alpha particles. The most stable atoms are those around the size of iron with a mass number in the mid fifties.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
